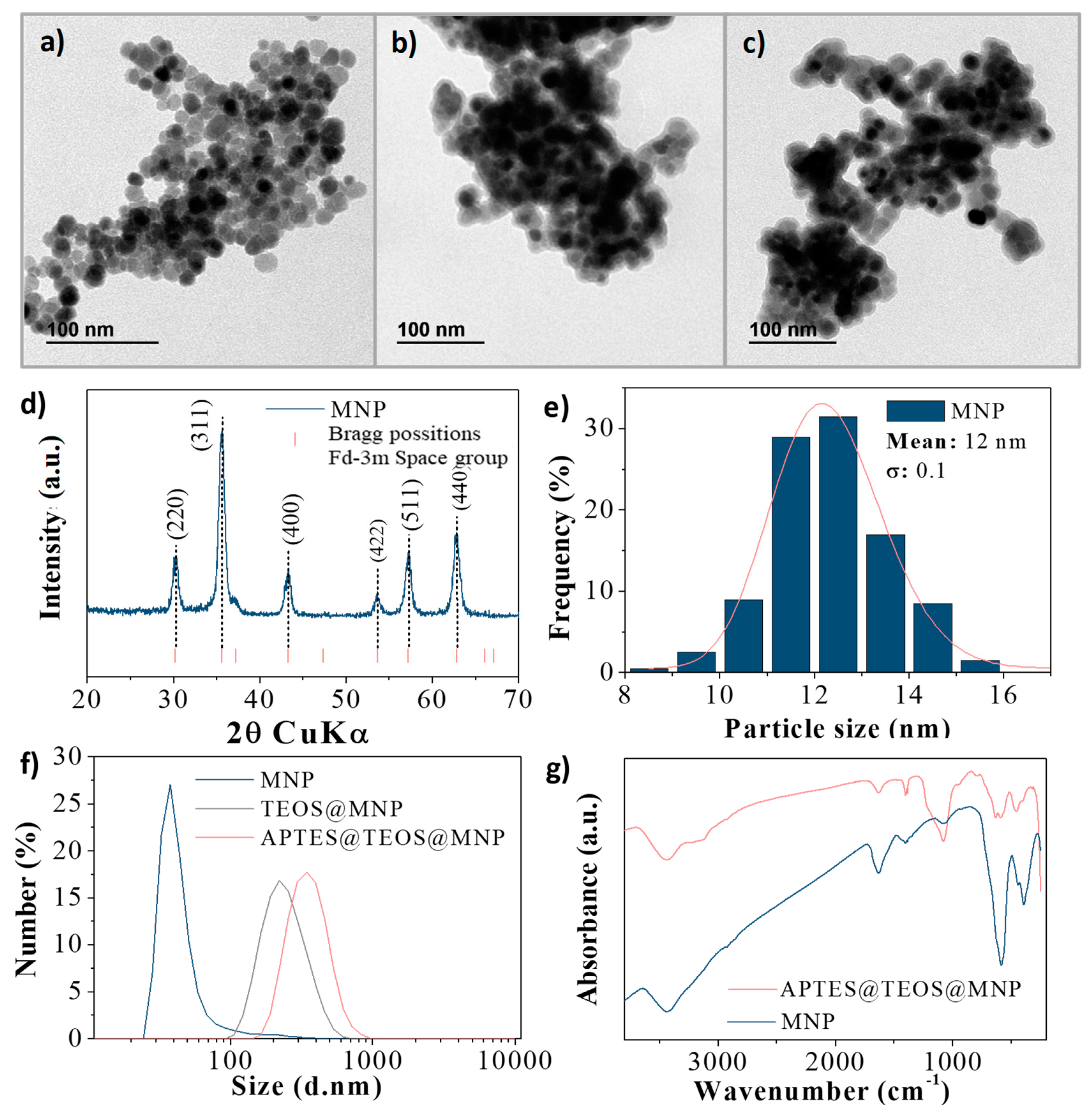
In such cases, it is necessary for the compounds to have different names that distinguish among the possible arrangements. For example, saying “C-A-three-P-O-four-two” for Ca 3(PO 4) 2 is much more difficult than saying “calcium phosphate.” In addition, you will see in Section 2.4 "Naming Covalent Compounds" that many compounds have the same empirical and molecular formulas but different arrangements of atoms, which result in very different chemical and physical properties. First, they are inconvenient for routine verbal communication. The chromate, CrO42, and dichromate, Cr2O72, ions are the most important species formed by chromium in the 6 oxidation state.The empirical and molecular formulas discussed in the preceding section are precise and highly informative, but they have some disadvantages. However, there are a few stable compounds of the states +5, 4, and +1. What is chromium’s oxidation state?Ĭhromium’s most common oxidation states are 6, 3, and 2. Valence electrons are found on an atom’s outermost shell, or energy level. What is the value of chromium?Ĭhromium has six valence electrons, according to the answer and explanation. Insulin action and glucose breakdown are also aided by chlorium. They are necessary for brain function and other bodily functions. It stimulates the synthesis of fatty acids and cholesterol. Martin Heinrich Klaproth Louis Nicolas Vauquelin What is the significance of chromium in the human body?Ĭhromium is required for fat and carbohydrate breakdown. 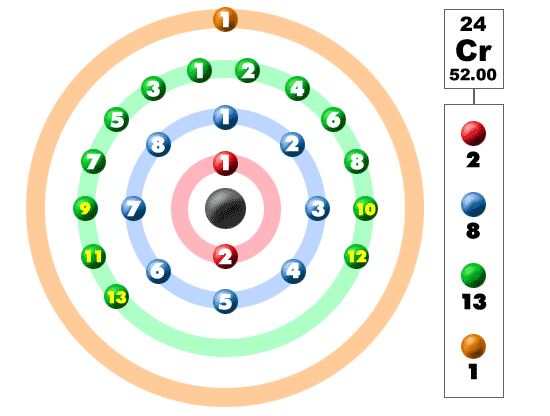
Chromium is also a blue-gray color with high melting and boiling points, as well as a good conductor of heat. All of those elements (including chromium) are non-reactive, good electrical conductors, shiny, and have a tough texture. On the Periodic Table (of Elements), chrome is the transition metals group. How many unpaired electrons are there in an atom of chromium? What is chromium in?ġ797 Is chromium an excellent electrical conductor?.What is the formula for chromium VI phosphide?.What is the formula for chromium VI sulfide?.Chromium (III) is a necessary nutrient in humans. Natural or industrial sources of chromium can be used to expose people.Ĭhromium III is far less toxic than chromium (VI). Trivalent chromium (Cr III) and hexavalent chromium (Cr VI) are the most common valence states in the environment. What’s the difference between chromium 3 and chrome 6? 
As a result, as in iron, which is also a transition metal, the valencies of chromium can be calculated as 2 and 3. What is the value of chromium 3?Ĭhromium has two oxidation states: 2 and 3, and their chemical formulas are CrCl2 and CrCl3. Chromium III Cr3 and Chromium VI Cr6 are the most stable, with the latter being a powerful oxidizing agent that is toxic and carcinogenic. How many electrons does a 5224cr3 Chromium III ion have? It can form a variety of ions as a Group 6 transition metal with six valence electrons in the outer shell (3d5, 4s1). The total negative charge is 6 if you take the number of O 2-ions. The total positive charge is 6 if you take the number of Cr 3 ions. Two Cr 3 ions and three O 2- ions make up a Cr*2O3*. Number Element Charge 24 Chromium 2, 3, 6, 25 manganese 2, 4, 7, 26 iron 2, 3, 27 cobalt 2, 3 chromium 2, 3 chromium 2, 3 chromium 2, 3 chromium 2, 3 chromium 2, 3 chromium 2, 3 chromium 2, 3 chromium 2, 3 chrom PubChem – Chromium(III) | Cr3 People also wonder what the charge of chromium in Cr2O3 is. Chromium(III) ion Cr3 5 Reactions use: Chromium(III) ion 11 Reactions form: Chromium(III) ion Cr 1 Charge = 3 Real ion What is the charge of chromium, people wonder? Gram formula weight (molecular mass) = 51.996.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
